Surgical resection of the early-stage localized disease foci is the only treatment modality for malignant melanoma. Metastatic melanoma is usually resistant to the standard cytotoxic therapy, including highly toxic drug combinations. Hence, immunotherapy has become the mainstay of treatment in advanced melanoma.
We evaluated the overall 3-year survival rate in 32 vaccine-treated patients with stage VI disease. Their clinical characteristics are presented in Table 1. All patients from the treatment group had a confirmed disease diagnosis. The control group was composed retrospectively to combine patients who received conventional therapy (prognostic and clinical characteristics of patients from control and treatment groups were matched). Throughout the follow-up period patients from the treatment group received no other systemic therapy other than immunotherapy. If clinically indicated, patients from the treatment and control groups underwent cytoreductive palliative surgery.
Table 1. Clinical characteristics of patients.
| Characteristic | Treatment | Control |
| Number of patients | 32 | 32 |
| Males/females | 10/22 | 10/22 |
| Age, years (median, range) | 48.8 (18-69) | 48.2 (24-77) |
| Site of metastases | ||
| Lymph node, skin/soft tissue | 23 (70 %) | 26 (81 %) |
| Lung | 10 (31 %) | 6 (19 %) |
| Liver | 7 (22 %) | 7 (22%) |
| Other organs | 7 (21 %) | 8 (25 %) |
| Prior treatment: | ||
| Surgery | 17 (53 %) | 16 (50 %) |
| Surgery + chemotherapy | 9 (28 %) | 10 (31 %) |
| Surgery + immunotherapy (IFN) | 0 (0 %) | 1 (3 %) |
| Surgery + chemotherapy + immunotherapy (IFN) | 2 (6 %) | 2 (6 %) |
| Surgery + chemotherapy + physiotherapy | 2 (6 %) | 0 (0 %) |
| Surgery + chemotherapy + radiotherapy | 1 (3 %) | 0 (0 %) |
| No treatment | 1 (3 %) | 3 (9 %) |
The results obtained suggest clinical safety and low toxicity (much lower compared with standard therapy for melanoma) of the vaccine-based therapy. No evidence of vaccine-mediated systemic autoimmune disorders was obtained, which could theoretically develop owing to the polyantigenic nature of the cellular vaccine used.
As shown in Figure 1, median survival rate in the vaccine-treated patients was significantly longer (P < 0.05) than that in the control patients. The overall 3-year survival rate in vaccine-treated and control patients was 25% and 2%, respectively. Clinical responses of various degrees of more than 6 months’ duration was observed in 21/32 (66%) of patients from the treatment group: complete response, partial response and disease stabilization was achieved in 5 (16%), 2 (6%) and 14 (44%) patients, respectively. Hence, xenovaccinotherapy could markedly prolong survival in a significant subset of advanced melanoma patients.
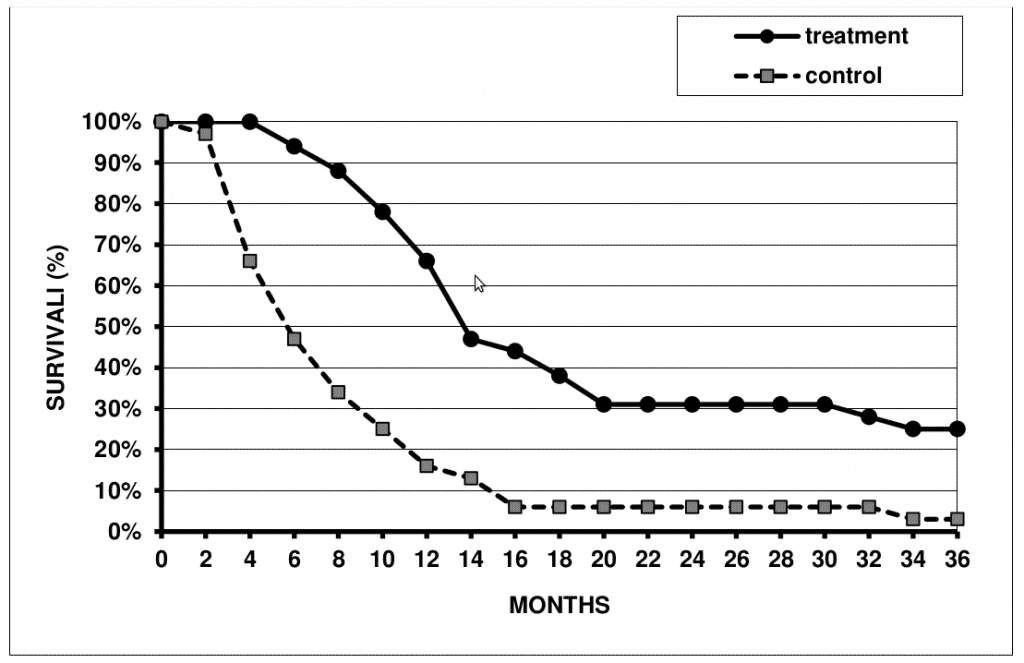
Figure 1. Survival rate in XCV-treated and control patients.
Importantly, almost all XСV-vaccinated patients who survived for 3 years after immunotherapy onset survived for 2 more years and longer. According to our data, 5- year overall survival rate in 51 XPV – treated patients with IV stage disease was about 20%. It should be noted that patients with very advanced disease (most of them with an untreatable metastatic process) were subjected to vaccine therapy in this study.
We believe that maximal immunotherapy efficiency can be achieved when applied in preoperative or postoperative manner, i.e. before or immediately after surgical resection of primary tumor and regional metastases. Consistent with this notion, our data indicates that survival of patients with III stage melanoma who received postoperatively vaccine therapy was significantly higher compared to that of control patients (Figure 2).
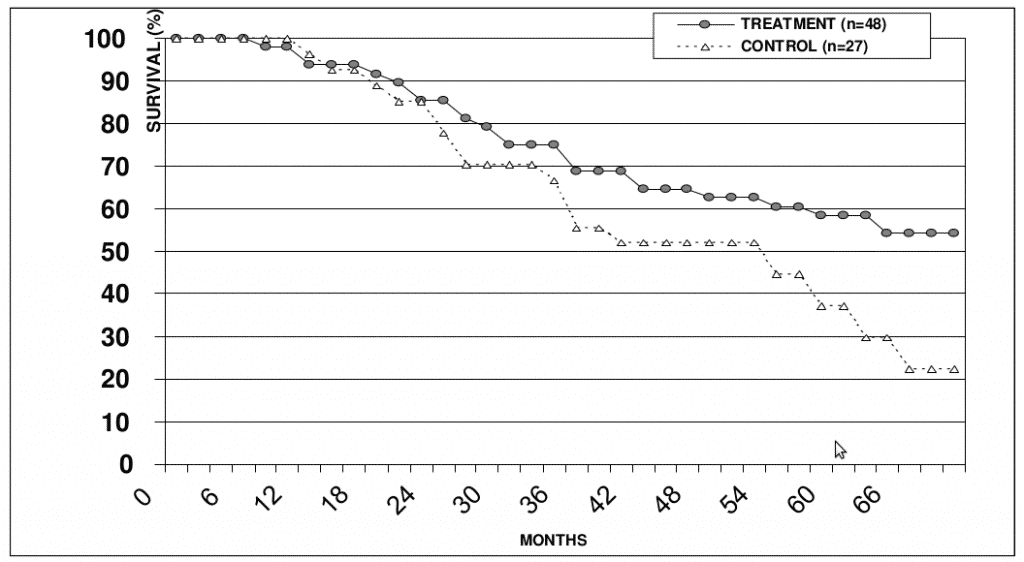
Figure 2. Survival rates in patients with III stage melanoma from the treatment and control groups.
Thus, the results suggest that xenovaccinotherapy could be effectively applied for prophylaxis of melanoma relapse in surgically treated patients. Since this treatment has no serious side effects, its clinical application could be advised in patients with I-III stages melanoma.
Examples:
Case 1. A 25 year-old female patient was referred to our Centre due to ulceration of nevus pigmentosus of the anterior chest. Upon surgery, epithelioid cell melanoma (III stage of Klark invasion; penetration depth – 2 mm) was diagnosed histologically. Two months later, ultrasonography revealed an enlargement of axillary lymph nodes (15 mm) on the left, which were surgically removed, and the presence of melanoma cells was confirmed histologically. Three months later, MRI showed multiple lesions (18 mm) and enlarged lymph nodes in the mediastinum. Two cycles of high level general controlled hyperthermia (43.30С) was used; however lesions in the liver and the mediastinum remained and had a tendency to enlarge, as judged from MRI data. The patient developed profound general weakness. Relative lymphocytosis (41%) was noted in the blood test. Vaccine therapy was initiated, and one day after each cell administrationan increase in body temperature to 380С and swelling around an injection site (~15 cm) were registered. MRI revealed no signs of metastatic lesions in the liver at 4 months follow-up (Figure 3 A,B). The remission lasted for 11 months, after which disease progression was noted in the form of multiple subcutaneous lesions on the mammary glands and the left upper arm . All metastatic lesions were surgically removed, and a post-operative vaccination course was applied. The subsequent remission lasted for 10 months, after which subcutaneous lesions appeared in the right lumbar region. Those lesions were surgically removed. Combined treatment (surgery + vaccine therapy) ultimately provided complete long-term remission. Final follow-up 3.5 years after vaccine therapy onset found the patient in good condition with no signs of disease.
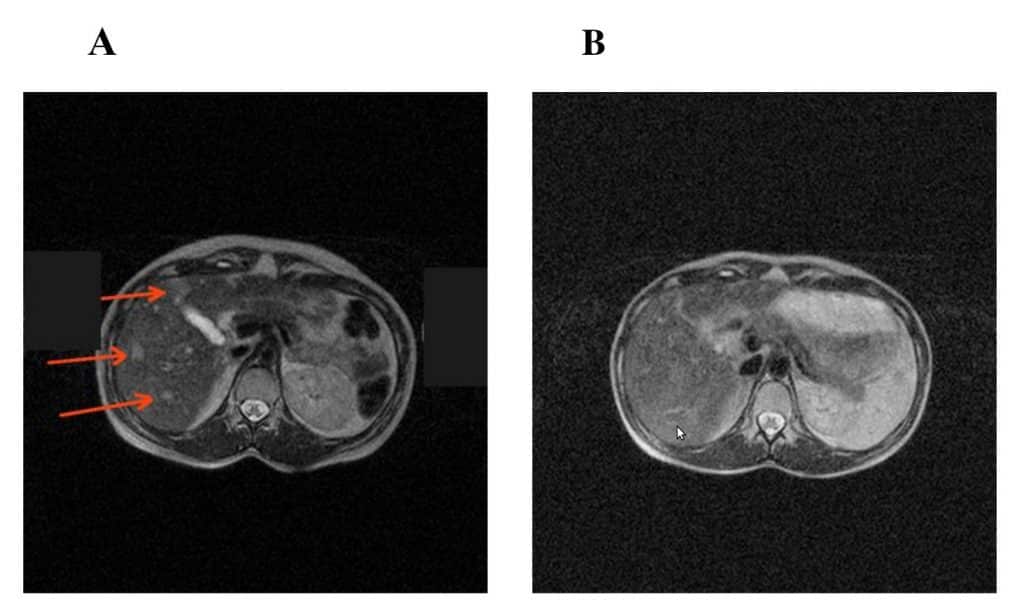
Figure 3. MRI scan of patient P before (A) and at 6 months (B) after vaccinotherapy initiation. The metastatic lesions are indicated by pointers.
Case 2. A 69 year-old female patient was diagnosed with skin melanoma lesions on the right foot, inguinal lymph nodes and multiple focal 5-6 mm lesions in the right lobe of the liver (VII segment) (by ultrasonography). Vaccine therapy was initiated after resection of primary tumor and affected lymph nodes. At that time, the patient complained of periodic rises in body temperature, marked general weakness, and lymphostasis in the right leg. Eight months later, ultrasonography revealed several small calcified foci in both liver lobes without signs of growth. One year later, ultrasonography revealed multiple hypoechoic 17 mm lesions in the right liver lobe, disseminated 2 mm lesions in the liver, and 13 mm lesions in the abdominal cavity. A vaccine therapy course was applied. One year later, the patient’s status was satisfactory. Multiple calcified 8 mm foci were revealed by ultrasonography. Final 4 year follow-up showed no evidence for disease progression.
From these examples it appears that the progression of the disease should not be a reason for discontinuation of immunotherapy.
Related scientific publications:
- A phase I vaccination study with dendritic cells loaded with NY-ESO-1 and α-galactosylceramide: induction of polyfunctional T cells in high-risk melanoma patients.
Gasser O, Sharples KJ, Barrow C, Williams GM, Bauer E, Wood CE, Mester B, Dzhelali M, Caygill G, Jones J, Hayman CM, Hinder VA, Macapagal J, McCusker M, Weinkove R, Painter GF, Brimble MA, Findlay MP, Dunbar PR, Hermans IF. Cancer Immunol Immunother. 2017 Nov 1. doi: 10.1007/s00262-017-2085-9. [Epub ahead of print]
https://www.ncbi.nlm.nih.gov/pubmed/29094183 - Effective tumor immunity to melanoma mediated by B16F10 cancer stem cell vaccine.
Zhao F, Zhang R, Wang J, Wu D, Pan M, Li M, Guo M, Dou J. Int Immunopharmacol. 2017 Nov;52:238-244. doi: 10.1016/j.intimp.2017.09.019. Epub 2017 Sep 23.
https://www.ncbi.nlm.nih.gov/pubmed/28950176 - Advances in Immunotherapy for Melanoma: A Comprehensive Review.
Rodríguez-Cerdeira C, Gregorio MC, López-Barcenas A, Sánchez-Blanco E, Sánchez-Blanco B, Fabbrocini G, Bardhi B, Sinani A, Guzman RA. Mediators Inflamm. 2017;2017:3264217. doi: 10.1155/2017/3264217. Epub 2017 Aug 1.
https://www.ncbi.nlm.nih.gov/pubmed/28848246 - Enhancing Immune Responses to Cancer Vaccines Using Multi-Site Injections.
Mould RC, AuYeung AWK, van Vloten JP, Susta L, Mutsaers AJ, Petrik JJ, Wood GA, Wootton SK, Karimi K, Bridle BW. Sci Rep. 2017 Aug 16;7(1):8322. doi: 10.1038/s41598-017-08665-9.
https://www.ncbi.nlm.nih.gov/pubmed/28814733 - An immunogenic personal neoantigen vaccine for patients with melanoma.
Ott PA, Hu Z, Keskin DB, Shukla SA, Sun J, Bozym DJ, Zhang W, Luoma A, Giobbie-Hurder A, Peter L, Chen C, Olive O, Carter TA, Li S, Lieb DJ, Eisenhaure T, Gjini E, Stevens J, Lane WJ, Javeri I, Nellaiappan K, Salazar AM, Daley H, Seaman M, Buchbinder EI, Yoon CH, Harden M, Lennon N, Gabriel S, Rodig SJ, Barouch DH, Aster JC, Getz G, Wucherpfennig K, Neuberg D, Ritz J, Lander ES, Fritsch EF, Hacohen N, Wu CJ. Nature. 2017 Jul 13;547(7662):217-221. doi: 10.1038/nature22991. Epub 2017 Jul 5.
https://www.ncbi.nlm.nih.gov/pubmed/28678778
