Colorectal carcinoma is one of the most common cancers. Surgical resection of early-stage localized disease is the only treatment modality. Advanced colon cancer is usually resistant to cytotoxic therapy including chemotherapy and radiotherapy. On the other hand, evidence is accumulated that immune-based approaches could significantly affect the course of disease.
We performed a case-control study to evaluate the overall 2-year survival rate in 37 vaccine-treated colorectal cancer patients ( clinical characteristics are presented in Table 1). All patients from the treatment group had a confirmed disease diagnosis, and the control group was composed retrospectively to combine patients who received conventional therapy (prognostic and clinical characteristics of patients from control and treatment groups were matched). Throughout the follow-up period, patients from the treatment group received no systemic therapy other than immunotherapy.
Table 1. Clinical characteristics of patients recruited in the case-control study.
| Characteristic | Treatment | Control |
| Number of patients | 37 | 37 |
| Males/females | 20/17 | 20/17 |
| Age, years (median, range) | 61.1 (38-79) | 48.2 (30-80) |
| Site of metastases | ||
| Lymph node, skin/soft tissue | 17 (46 %) | 17 (46 %) |
| Lung | 8 (22 %) | 6 (16 %) |
| Liver | 27 (73 %) | 19 (51 %) |
| Other organs | 11 (30 %) | 8 (22 %) |
| Prior treatment: | ||
| Surgery | 19 (51 %) | 17 (46 %) |
| Surgery + chemotherapy | 9 (24 %) | 8 (22 %) |
| Surgery + radiotherapy | 3 (8 %) | 5 (13 %) |
| Surgery + chemotherapy + radiotherapy | 1 (3 %) | 1 (3 %) |
| Radiotherapy | 1 (3 %) | 1 (3 %) |
| No treatment | 4 (11 %) | 5 (13 %) |
As shown in Figure 1, the median survival rate in XPV(xenogeneic polyantigenic vaccine) -treated patients was significantly longer (P < 0.05), compared to that in control patients (17 vs. 7 months). The overall 2-year survival rate in XPV-treated and control group was 27% (10 patients) and 3% (1 patient), respectively. Clinical effects of various degrees (complete response, partial response and disease stabilization) with duration of ≥6 months was observed in 23/37 (62%) of patients from the treatment group.
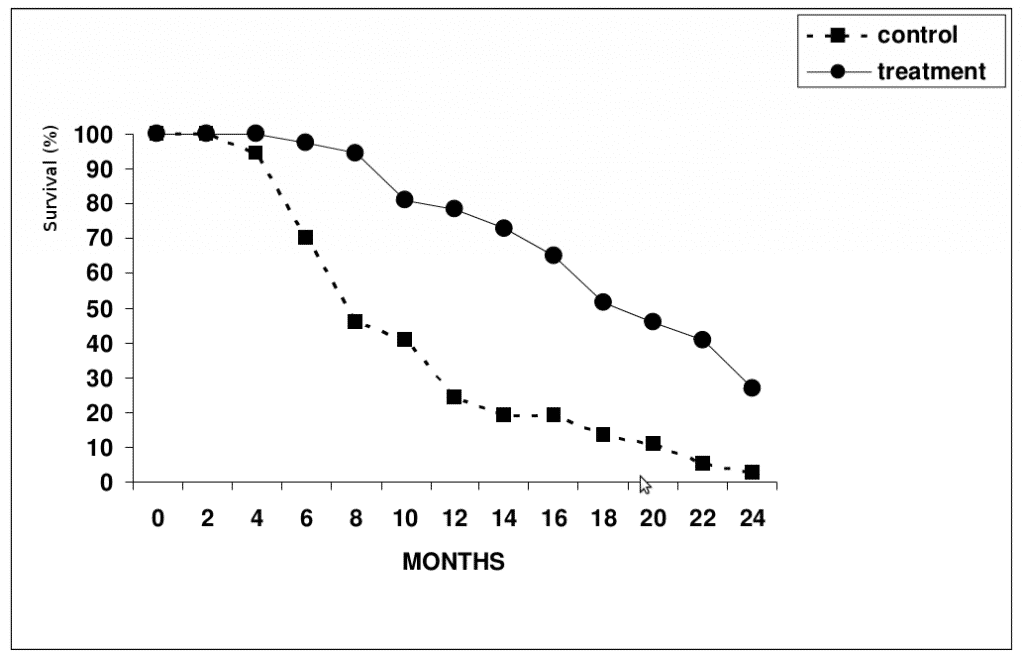
Figure 1. Survival rate in patients from the control (n=37) and XCV-treated (n=37) groups .
Importantly, this study included patients with very advanced disease. We envisage that XVT is most effective when initiated before or immediately after surgical resection of the primary tumor and its regional metastases.
Examples.
Case 1. A 51 year-old female patient was admitted to the surgery department with acute bowel obstruction During laparotomy tumor conglomerates were noted in the sigmoid colon, in the retroperitoneal space and in the left uterine appendages. In addition, abscess of the mesocolon was found. Cytoreductive surgery with a colostomy was performed. A mucus-producing adenocarcinoma was diagnosed by histological analysis. A vaccine therapy course was initiated after a patient’s discharge from the hospital. The treatment was well tolerated. Secondary liver tumor lesions (up to 25 mm) were detected 10 months after the immunotherapy onset. Ultrasonography confirmed a 34 x 27 mm solid mass in the left retroperitoneal space. Nevertheless, we continued an immunotherapy of the patient r. One year later, ultrasonography and computerized tomography (CT) revealed no signs of metastatic lesions in the liver, and anastomosis/colostomy closure was performed. Final follow-up 3 years after the onset of vaccine therapy found the patient in good condition without serious complains.
Case 2. A 46 year-old male patient underwent surgery for colon cancer. A low-grade differentiated adenocarcinoma was diagnosed by histological examination. A focal 15 mm liver lesion was found by ultrasonography and CT. A vaccine therapy course was initiated, and no secondary liver lesions were detected by ultrasonography 1.5 years after the immunotherapy onset in June 2001. A 4 years follow-up showed that the patient was in good condition without any signs of disease.
Related scientific publications:
- Immunotherapy regimens for metastatic colorectal carcinomas.
Bashir B, Snook AE. Hum Vaccin Immunother. 2017 Oct 30:0. doi: 10.1080/21645515.2017.1397244. [Epub ahead of print]
https://www.ncbi.nlm.nih.gov/pubmed/29083978 - The emerging role of immunotherapy in colorectal cancer.
Lynch D, Murphy A. Ann Transl Med. 2016 Aug;4(16):305. doi: 10.21037/atm.2016.08.29. Review.
https://www.ncbi.nlm.nih.gov/pubmed/27668225 - Immunotherapy in human colorectal cancer: Challenges and prospective.
Sun X, Suo J, Yan J. World J Gastroenterol. 2016 Jul 28;22(28):6362-72. doi: 10.3748/wjg.v22.i28.6362. Review.
https://www.ncbi.nlm.nih.gov/pubmed/27605872 - Immunotherapy in colorectal cancer: What have we learned so far?
Sanchez-Castañón M, Er TK, Bujanda L, Herreros-Villanueva M. Clin Chim Acta. 2016 Sep 1;460:78-87. doi: 10.1016/j.cca.2016.06.027. Epub 2016 Jun 24. Review.
https://www.ncbi.nlm.nih.gov/pubmed/27350293 - Immunotherapy of Metastatic Colorectal Cancer: Prevailing Challenges and New Perspectives.
Zumwalt TJ, Goel A. Curr Colorectal Cancer. Rep. 2015 Jun 1;11(3):125-140. Epub 2015 Jun 29.
https://www.ncbi.nlm.nih.gov/pubmed/26441489
